Share
The iodide (I2) content of a commercial mineral water was measured by two methods that produced wildly different results.6 Method A found 0.
Question
The iodide (I2) content of a commercial mineral water was measured by two methods that produced wildly different results.6 Method A found 0.23 milligrams of I2 per liter (mg/L) and method B found 0.009 mg/L. When Mn21 was added to the water, the I2 content found by method A increased each time that more Mn21 was added, but results from method B were unchanged. Which of the Terms to Understand describes what is occurring in these measure- ments? Which result is more reliable?
in progress
0
Chemistry
5 years
2021-07-10T19:05:41+00:00
2021-07-10T19:05:41+00:00 1 Answers
313 views
1

Answers ( )
Answer:
Results from method B is more reliable than method A.
Explanation:
The two method that are used for the analysis produced different results. The first method that is method A gives higher value of the iodine content than the method B.
When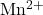 was added to water, method A showed an increased in the iodine content and it increases with the increase in the amount of
was added to water, method A showed an increased in the iodine content and it increases with the increase in the amount of 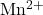 .
.
Where as in the method B, there is no change in the results. Therefore the measurements provided by the method A shows an inference of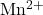 ion.
ion.
The measurement of the iodine content is affected by the presence of the ion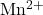 in water.
in water.
Since in method B there is no change in measurement, it is independent of the presence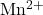 ion in water.
ion in water.
As higher iodine content is given by method A, so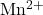 ion must be present in original water that must be interfering the measurement. Hence, method B is more reliable.
ion must be present in original water that must be interfering the measurement. Hence, method B is more reliable.