Calculate the number of hydrogen atoms present in 40g of urea, (NH2)2CO Question Calculate the number of hydrogen atoms present in 40g of urea, (NH2)2CO in progress 0 Chemistry Doris 5 years 2021-08-02T12:43:43+00:00 2021-08-02T12:43:43+00:00 1 Answers 258 views 0
Answers ( )
Answer: There are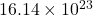 atoms of hydrogen are present in 40g of urea,
atoms of hydrogen are present in 40g of urea, 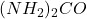 .
.
Explanation:
Given: Mass of urea = 40 g
Number of moles is the mass of substance divided by its molar mass.
First, moles of urea (molar mass = 60 g/mol) are calculated as follows.
According to the mole concept, 1 mole of every substance contains atoms.
atoms.
So, the number of atoms present in 0.67 moles are as follows.
In a molecule of urea there are 4 hydrogen atoms. Hence, number of hydrogen atoms present in 40 g of urea is as follows.
Thus, we can conclude that there are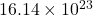 atoms of hydrogen are present in 40g of urea,
atoms of hydrogen are present in 40g of urea, 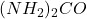 .
.