Share
Calcium carbonate is often used as an antacid. Your stomach acid is composed of HCl at a pH of 1.5. If you ate toooo much Turkey and need to
Question
Calcium carbonate is often used as an antacid. Your stomach acid is composed of HCl at a pH of 1.5. If you ate toooo much Turkey and need to neutralize 15.0 mL of stomach acid, how many grams of calcium carbonate would you need to take
in progress
0
Chemistry
5 years
2021-07-26T15:31:52+00:00
2021-07-26T15:31:52+00:00 1 Answers
27 views
0

Answers ( )
Answer: 0.0237 g of calcium carbonate would be required to neutralize the given amount of HCl
Explanation:
pH is defined as the negative logarithm of hydrogen ion concentration present in the solution
Given value of pH = 1.5
Putting values in equation 1:
Molarity is defined as the amount of solute expressed in the number of moles present per liter of solution. The units of molarity are mol/L. The formula used to calculate molarity:
We are given:
Volume of solution = 15.0 mL
Molarity of HCl = 0.0316 M
Putting values in equation 2:
The chemical equation for the reaction of HCl and calcium carbonate follows:
By the stoichiometry of the reaction:
2 moles of HCl reacts with 1 mole of calcium carbonate
So,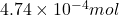 of HCl will react with =
of HCl will react with = 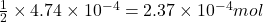 of calcium carbonate
of calcium carbonate
The number of moles is defined as the ratio of the mass of a substance to its molar mass.
Moles of calcium carbonate =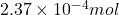
Molar mass of calcium carbonate = 100.01 g/mol
Putting values in the above equation:
Hence, 0.0237 g of calcium carbonate would be required to neutralize the given amount of HCl