Share
A gas of certain mass occupies volume of 1.2 L at 37°C and 3 atm. At what temperature will the volume and pressure of this gas become one-th
Question
Lost your password? Please enter your email address. You will receive a link and will create a new password via email.
Answers ( )
Answer: At a temperature of 34.44 K the volume and pressure of this gas become one-third of their initial values.
Explanation:
Given : = 1.2 L,
= 1.2 L, 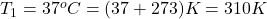
Formula used to calculate the final temperature is as follows.
Substitute the values into above formula as follows.
Thus, we can conclude that the final temperature is 34.44 K.