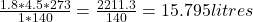Share
A gas at 1.8 atm, 140 K, and 4.5 L is adjusted to standard temperature and pressure conditions. What is the new volume?
Question
Lost your password? Please enter your email address. You will receive a link and will create a new password via email.
Answers ( )
From the equation,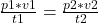 , we make v2 the subject of the formula, so that gives us:
, we make v2 the subject of the formula, so that gives us: 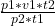
Standard temperature = 273k
Standard pressure = 1atm
Plugging in our values,