Share
3. A 3.455-g sample of a mixture was analyzed for barium ion by adding a small excess of sulfuric acid to an aqueous solution of the sample.
Question
3. A 3.455-g sample of a mixture was analyzed for barium ion by adding a small excess of sulfuric acid to an aqueous solution of the sample. The resultant reaction produced a precipitate of barium sulfate, which was collected by filtration, washed, dried, and weighed. If 0.2815 g of barium sulfate was obtained, what was the mass percentage of barium in the sample
in progress
0
Chemistry
5 years
2021-07-13T08:38:26+00:00
2021-07-13T08:38:26+00:00 1 Answers
84 views
0

Answers ( )
Answer:
Explanation:
From the question we are told that:
Mass of mixture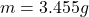
Mass of Barium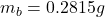
Equation of Reaction is given as
Generally the equation for Moles of Barium is mathematically given by
Since
Therefore
Generally the equation for Mass of Barium is mathematically given by
Therefore