Share
In the coal-gasification process, carbon monoxide is converted to carbon dioxide via the following reaction: CO (g) H2O (g) CO2 (g) H2 (
Question
In the coal-gasification process, carbon monoxide is converted to carbon dioxide via the following reaction: CO (g) H2O (g) CO2 (g) H2 (g) In an experiment, 0.35 mol of CO and 0.40 mol of H2O were placed in a 1.00-L reaction vessel. At equilibrium, there were 0.19 mol of CO remaining. Keq at the temperature of the experiment is __________. A) 5.47 B) 1.0 C) 1.78 D) 0.75 E) 0.56
in progress
0
Chemistry
5 years
2021-08-15T22:57:39+00:00
2021-08-15T22:57:39+00:00 1 Answers
360 views
1

Answers ( )
Answer: at the temperature of the experiment is 0.56.
at the temperature of the experiment is 0.56.
Explanation:
Moles of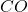 = 0.35 mole
= 0.35 mole
Moles of = 0.40 mole
= 0.40 mole
Volume of solution = 1.00 L
Initial concentration of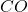 =
= 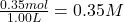
Initial concentration of =
= 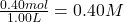
Equilibrium concentration of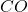 =
= 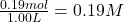
The given balanced equilibrium reaction is,
Initial conc. 0.35 M 0.40 M 0 M 0M
At eqm. conc. (0.35-x) M (0.40-x) M (x) M (x) M
Given: (0.35-x) = 0.19
x= 0.16 M
The expression for equilibrium constant for this reaction will be,
Now put all the given values in this expression, we get :
Thus at the temperature of the experiment is 0.56.
at the temperature of the experiment is 0.56.