Share
Helium gas contained in a piston cylinder assembly undergoes an isentropic polytropic process from the given initial state with P1 = 0.02 ba
Question
Helium gas contained in a piston cylinder assembly undergoes an isentropic polytropic process from the given initial state with P1 = 0.02 bar, T1 = 200 K to the final state with P2 = 0.14 bar. Determine the work and heat transfer (per unit mass) involved in this process.
in progress
0
Physics
5 years
2021-08-22T23:39:03+00:00
2021-08-22T23:39:03+00:00 1 Answers
12 views
0

Answers ( )
Answer:
Heat transfer during the process = 0
Work done during the process = – 371.87 KJ
Explanation:
Initial pressure = 0.02 bar
= 0.02 bar
Initial temperature = 200 K
= 200 K
Final pressure = 0.14 bar
= 0.14 bar
Gas constant for helium R = 2.077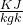
This is an isentropic polytropic process so temperature – pressure relationship is given by the following formula,
Put all the values in above formula we get,
⇒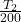 =
= ![Rendered by QuickLaTeX.com [\frac{0.14 }{0.02 } ]^{\frac{1.4 - 1}{1.4} }](https://documen.tv/wp-content/ql-cache/quicklatex.com-7954438ae2f33a17b30c46bf7d0c9d61_l3.png)
⇒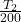 = 1.74
= 1.74
⇒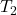 = 348.72 K
= 348.72 K
This is the final temperature of helium.
For isentropic polytropic process heat transfer to the system is zero.
⇒ ΔQ = 0
Work done W = m × ( –
– 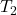 ) ×
) × 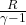
⇒ W = 1 × ( 200 – 348.72 ) ×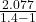
⇒ W = 371.87 KJ
This is the work done in this process. here negative sign shows that work is done on the gas in the compression of gas.