Share
When 2.935 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 9.920 grams of CO2 and 2.031 grams of H2O were prod
Question
When 2.935 grams of a hydrocarbon, CxHy, were burned in a combustion analysis apparatus, 9.920 grams of CO2 and 2.031 grams of H2O were produced. In a separate experiment, the molar mass of the compound was found to be 26.04 g/mol. Determine the empirical formula and the molecular formula of the hydrocarbon.
in progress
0
Chemistry
5 years
2021-07-22T22:23:02+00:00
2021-07-22T22:23:02+00:00 1 Answers
16 views
0

Answers ( )
Answer: The empirical and molecular formula for the given organic compound are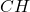 and
and 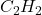 respectively.
respectively.
Explanation:
The empirical formula is the chemical formula of the simplest ratio of the number of atoms of each element present in a compound.
The chemical equation for the combustion of hydrocarbon having carbon and hydrogen follows:
where, ‘x’ and ‘y’ are the subscripts of carbon and hydrogen respectively.
We are given:
Mass of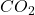 = 9.920 g
= 9.920 g
Mass of = 2.031 g
= 2.031 g
In 44 g of carbon dioxide, 12 g of carbon is contained.
So, in 9.920 g of carbon dioxide,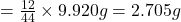 of carbon will be contained.
of carbon will be contained.
In 18g of water, 2 g of hydrogen is contained.
So, in 2.031 g of water,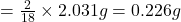 of hydrogen will be contained.
of hydrogen will be contained.
The number of moles is defined as the ratio of the mass of a substance to its molar mass. The equation used is:
To formulate the empirical formula, we need to follow some steps:
Molar mass of C = 12 g/mol
Molar mass of H = 1 g/mol
Putting values in equation 1, we get:
Calculating the mole fraction of each element by dividing the calculated moles by the least calculated number of moles that is 0.225 moles
The ratio of C : H = 1 : 1
The empirical formula of the compound becomes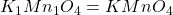
To calculate the molecular formula, the number of atoms of the empirical formula is multiplied by a factor known as valency that is represented by the symbol, ‘n’.
We are given:
Mass of molecular formula = 26.04 g/mol
Mass of empirical formula = 13 g/mol
Putting values in equation 3, we get:
Multiplying this valency by the subscript of every element of empirical formula, we get:
Hence, the empirical and molecular formula for the given organic compound are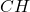 and
and 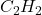 respectively.
respectively.