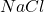Share
An Element X has been discovered. Element X forms an Xt ion and G9 EOT3 2020- has an atomic number of 11. The X* ion of Element
Question
An Element X has been discovered. Element X forms an Xt ion and
G9 EOT3 2020-
has an atomic number of 11. The X* ion of Element X forms a bond
with CI.
1-3
5.00
11
Na
Mg
3
22.99
19
24.10
20
21
K
Ca
Sc
19.10
400
19
Rb
Sr
Y
NOT
R$ 47
53
N7
so
CS
Ba
3.71
112.91
117 11
Ra
malleable and co
malene bi
in progress
0
Chemistry
5 years
2021-07-23T07:04:31+00:00
2021-07-23T07:04:31+00:00 1 Answers
15 views
0

Answers ( )
Answer: The chemical symbol of the compound formed is or
or 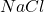
Explanation:
An ionic compound is formed when the complete transfer of electrons takes place from one element (usually metals) to another element (usually non-metals).
The element losing electron (metal) forms a positive ion known as a cation while the element gaining electron (non-metal) forms a negative ion known as an anion.
Element X has the atomic number 11 which is sodium. The electronic configuration of it is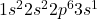
This element will lose 1 electron to form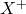 ion of
ion of 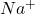
Chlorine has the atomic number 17. The electronic configuration of it is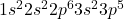
This element will gain 1 electron to form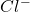 ion
ion
When these two ions combine, they lead to the formation of an ionic compound having a chemical formula of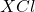 or
or 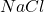
Hence, the chemical symbol of the compound formed is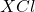 or
or