Share
A piece of corroded steel plate was found in a submerged ocean vessel. It was estimated that the original area of the plate was 11 in.2 and
Question
A piece of corroded steel plate was found in a submerged ocean vessel. It was estimated that the original area of the plate was 11 in.2 and that approximately 2.3 kg had corroded away during the submersion. Assuming a corrosion penetration rate of 200 mpy for this alloy in seawater, estimate the time of submersion in years. The density of steel is 7.9 g/cm3.
in progress
0
Physics
5 years
2021-07-23T18:39:38+00:00
2021-07-23T18:39:38+00:00 1 Answers
335 views
0

Answers ( )
Answer:
The time of the submersion is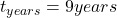
Explanation:
From the question we are told that
The area of the plate is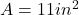
The mass of the corroded plate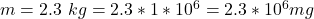
The corrosion penetration rate is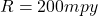
The density of steel is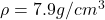
Generally the corrosion penetration rate can be mathematically represented as
Where K is the corrosion imperial unit constant whose value is K = 534 mpy
t is the exposure time of the plate
Making t the subject of the formula
Substituting value
Converting to years