5) How many Kilograms are in 12 L of Chlorine Gas? Question 5) How many Kilograms are in 12 L of Chlorine Gas? in progress 0 Chemistry Thu Thảo 5 years 2021-08-10T06:51:09+00:00 2021-08-10T06:51:09+00:00 1 Answers 12 views 0
Answers ( )
Answer: 0.03798 kilograms
Explanation:
According to avogadro’s law, 1 mole of every substance occupies 22.4 L at STP and contains avogadro’s number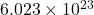 of particles.
of particles.
To calculate the moles, we use the equation:
1 mole of chlorine gas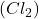 weighs = 71 g
weighs = 71 g
Thus 0.535 moles of chlorine gas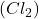 will weigh =
will weigh = 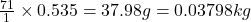 (1kg=1000g)
(1kg=1000g)
Thus there are 0.03798 kilograms in 12 L of chlorine gas.