Share
In the acetylene torch, acetylene gas (C2H2) burns in oxygen to produce carbon dioxide water and energy. How many moles of CO2 are formed fr
Question
In the acetylene torch, acetylene gas (C2H2) burns in oxygen to produce carbon dioxide water and energy. How many moles of CO2 are formed from the reaction with 1.20 moles of C2H2?
Given the following equation
2C2H2(g) + 502(g) = 4CO2(g) + 2H2O(g)
in progress
0
Chemistry
5 years
2021-07-29T17:49:49+00:00
2021-07-29T17:49:49+00:00 1 Answers
63 views
0

Answers ( )
Answer: 2.40 moles of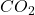 will be formed in the reaction.
will be formed in the reaction.
Explanation:
Given values:
Moles of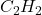 = 1.20 moles
= 1.20 moles
The given chemical equation follows:
By the stoichioemetry of the reaction:
If 2 moles of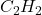 produces 4 moles of carbon dioxide
produces 4 moles of carbon dioxide
Then, 1.20 moles of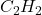 wil produce =
wil produce = 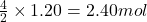 of carbon dioxide
of carbon dioxide
Hence, 2.40 moles of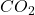 will be formed in the reaction.
will be formed in the reaction.