Share
In 8,450 seconds, the number of radioactive nuclei decreases to 1/16 of the number present initially. What is the half-life (in s) of the ma
Question
Lost your password? Please enter your email address. You will receive a link and will create a new password via email.
Answers ( )
Answer:
2113 seconds
Explanation:
The general decay equation is given as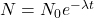 , then;
, then;
t is the time taken for decay to occur = 8,450s
Before we can find the half life of the material, we need to get the decay constant first.
Substituting the given values into the formula above, we will have;
Half life f the material is expressed as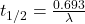
Hence, the half life of the material is approximately 2113 seconds