Share
Balance the following equation: AlCl3 + Cs –> CsCl + Al Group of answer choices A.2, 6, 6, 2 B.1, 3, 3, 1<
Question
Lost your password? Please enter your email address. You will receive a link and will create a new password via email.
Answers ( )
Answer: The balanced equation is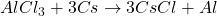
Explanation:
A chemical equation which contains same number of atoms on both reactant and product side is called a balanced chemical equation.
For example,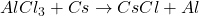
Here, number of atoms on reactant side are as follows.
Number of atoms on product side are as follows.
To balance this equation, multiply Cs by 3 on reactant side and multiply CsCl by 3 on product side.
Hence, now the equation will be as follows.
Here, number of atoms on reactant side are as follows.
Number of atoms on product side are as follows.
Thus, we can conclude that this equation is balanced.