Share
How does H3PO ionize in water ? Are these done correctly ? HNO3 → H + NO3 H2SO4 → H2 + SO4
Question
Lost your password? Please enter your email address. You will receive a link and will create a new password via email.
Answers ( )
Answer:
H3PO4 + H2O ↔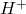 +
+ 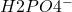
Explanation:
HNO3 → NO2 + H2O + O2
H2SO4 → SO3 + H2O + O2