Share
A compound that contains only carbon, hydrogen, and oxygen is 58.8% C and 9.87% H by mass. What is the empirical formula of this substance
Question
Lost your password? Please enter your email address. You will receive a link and will create a new password via email.
Answers ( )
Answer: The empirical formula of the compound becomes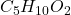
Explanation:
The empirical formula is the chemical formula of the simplest ratio of the number of atoms of each element present in a compound.
Let the mass of the compound be 100 g
Given values:
% of C = 58.8%
% of H = 9.87%
% of O = [100 – 58.8 – 9.87] = 31.33%
Mass of C = 58.8 g
Mass of H = 9.87 g
Mass of O = 31.33 g
The number of moles is defined as the ratio of the mass of a substance to its molar mass. The equation used is:
To formulate the empirical formula, we need to follow some steps:
Molar mass of C = 12 g/mol
Molar mass of H = 1 g/mol
Molar mass of O = 16 g/mol
Putting values in equation 1, we get:
Calculating the mole fraction of each element by dividing the calculated moles by the least calculated number of moles that is 1.96 moles
Converting the mole fraction into whole numbers by multiplying them with 2.
The ratio of C : H : O = 5 : 10 : 2
Hence, the empirical formula of the compound becomes